We report how the electron transport through a solid-state metal/Gly-Gly-His (GGH) tripeptide monolayer/metal junction and the metal/GGH work function (WF) are modified by the GGH complexation with Cu2+ ions. Conducting atomic force microscopy is used to measure the current-voltage histograms. The WF is characterized by combining macroscopic Kelvin probe and Kelvin probe force microscopy at the nanoscale. We observe that the complexation of Cu2+ions with the GGH monolayer is highly dependent on the molecular surface density and results in opposite trends. In the case of a high-density monolayer the conformational changes are hindered by the proximity of the neighboring peptides, hence forming an insulating layer in response to copper complexation. However, the monolayers of a slightly lower density allow for the conformational change to a looped peptide wrapping the Cu-ion, which results in a more conductive monolayer. Copper-ion complexation to the high- and low-density monolayers systematically induces an increase of the WFs. Copper-ion complexation to the low-density monolayer induces an increase of electron-transport efficiency, whereas the copper-ion complexation to the high-density monolayer results in a slight decrease of electron transport. Both of the observed trends agree with first-principle calculations. Complexation of copper to the low-density GGH monolayer induces a new gap state slightly above the Au Fermi energy that is absent in the high-density monolayer.
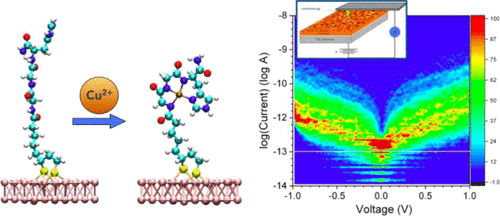
We report how the electron transport through a solid-state metal/Gly-Gly-His (GGH) tripeptide monolayer/metal junction and the metal/GGH work function (WF) are modified by the GGH complexation with Cu2+ ions. Conducting atomic force microscopy is used to measure the current-voltage histograms. The WF is characterized by combining macroscopic Kelvin probe and Kelvin probe force microscopy at the nanoscale. We observe that the complexation of Cu2+ions with the GGH monolayer is highly dependent on the molecular surface density and results in opposite trends. In the case of a high-density monolayer the conformational changes are hindered by the proximity of the neighboring peptides, hence forming an insulating layer in response to copper complexation. However, the monolayers of a slightly lower density allow for the conformational change to a looped peptide wrapping the Cu-ion, which results in a more conductive monolayer. Copper-ion complexation to the high- and low-density monolayers systematically induces an increase of the WFs. Copper-ion complexation to the low-density monolayer induces an increase of electron-transport efficiency, whereas the copper-ion complexation to the high-density monolayer results in a slight decrease of electron transport. Both of the observed trends agree with first-principle calculations. Complexation of copper to the low-density GGH monolayer induces a new gap state slightly above the Au Fermi energy that is absent in the high-density monolayer.
