Digital twin (DT) technology enables virtual replicas that support monitoring, simulation, and prediction for personalized care. A machine learning (ML)-driven DT framework for a field-effect transistor (FET)-based hormone biosensor targeting 17β-estradiol is presented, intended for future integration into a Smart Health Monitoring System (SHMS) for infertility treatment. Real-time biosensor recordings acquired under sequential estradiol exposures (0, 20, 150, 200, and 400 pg/mL) were processed using a protocol-aware pipeline. A row-level learning strategy was adopted, in which each timepoint was treated as an individual training sample and the normalized biosensor signal was enhanced with local temporal descriptors. Hormone concentration was formulated as a supervised regression target transformed as log10(ro + 1) and evaluated using leave-one-biosensor-out (LOBO) validation with out-of-fold (OOF) predictions. Among the evaluated models, CatBoost achieved the best LOBO performance (RMSElog = 0.240; R2 = 0.935), corresponding to an root mean squared error of 85.28 pg/mL in the original concentration domain. SHAP analysis indicated that prediction relied primarily on temporal and signal-morphology features, suggesting that physically meaningful response dynamics were learned rather than sensor-specific offsets. Finally, a one-step-ahead forward model was applied autoregressively to generate protocol-consistent synthetic Isd(t) signal, demonstrating the feasibility of DT-based signal simulation from real biosensor data. While the results are promising, validation on larger independent datasets and under physiologically complex conditions remains necessary to establish robustness and clinical readiness.
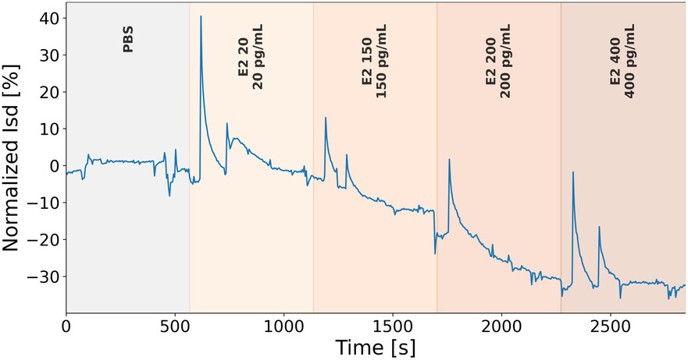
Digital twin (DT) technology enables virtual replicas that support monitoring, simulation, and prediction for personalized care. A machine learning (ML)-driven DT framework for a field-effect transistor (FET)-based hormone biosensor targeting 17β-estradiol is presented, intended for future integration into a Smart Health Monitoring System (SHMS) for infertility treatment. Real-time biosensor recordings acquired under sequential estradiol exposures (0, 20, 150, 200, and 400 pg/mL) were processed using a protocol-aware pipeline. A row-level learning strategy was adopted, in which each timepoint was treated as an individual training sample and the normalized biosensor signal was enhanced with local temporal descriptors. Hormone concentration was formulated as a supervised regression target transformed as log10(ro + 1) and evaluated using leave-one-biosensor-out (LOBO) validation with out-of-fold (OOF) predictions. Among the evaluated models, CatBoost achieved the best LOBO performance (RMSElog = 0.240; R2 = 0.935), corresponding to an root mean squared error of 85.28 pg/mL in the original concentration domain. SHAP analysis indicated that prediction relied primarily on temporal and signal-morphology features, suggesting that physically meaningful response dynamics were learned rather than sensor-specific offsets. Finally, a one-step-ahead forward model was applied autoregressively to generate protocol-consistent synthetic Isd(t) signal, demonstrating the feasibility of DT-based signal simulation from real biosensor data. While the results are promising, validation on larger independent datasets and under physiologically complex conditions remains necessary to establish robustness and clinical readiness.
